Exsil GFC 3000
High Performance Size Separation of Proteins
Size separation of proteins by Gel Filtration Chromatography (GFC) is widely used and the market-leading product in this area is the well-respected TSK G3000SWXL range.
The TSK product has good protein resolution due to its high pore volume but the down side of this is that the bed stability is compromised and the phase has a maximum operating pressure of 1000 psi. This imposes a limit on the speed of analysis and the ability to run multiple columns.
Exsil GFC 3000 has been developed to match the high efficiency of the TSK product and to give superior bed stability at an economical price.
- Phase: Stable High Polarity (pH 2-7.5)
- Base Silica: Exsil 3000S, high putity Silica
- Particle Size: 5 µm
- Pore Size: 235 A
- Pore Volume: 1.2 cc/g
This features:
- High Stability: Can be packed at 7000 psi
- High Efficiency: Typically 100 000 plates/m for 5µm
- High Recovery: No Evidence of protein loss
- Wide Applicability: Low absorption of basic proteins
- Cost Eddective: High performance at low cost
- Reproducible: Typical Exmere reliability
Size Exclusion Range
Conventional GPC of polystyrenes on Exsil GFC 3000 and TSK G3000SW using THF eluent, show the classic S shaped curve and a similar pore size distribution with the Exsil having a slightly greater population of large pores.
GFC of proteins shows a similar trend at higher molecular weights but increased retention of the lower molecular weight proteins on the TSK. These proteins are basic and it is possible that the extra retention is due to ionic interaction on the TSK. This culminates in the basic peptide Bacitracin being eluted after the apparent void volume.
Protein Separations
The following chromatograms illustrate the high efficiency achieved by these phases with over 110,000 plates/m for the Phenylalanine.. The Exsil shows improved resolution of the Thyroglobulin from the excluded peak while the TSK gives better resolution of Ovalbumin and Myoglobin but this is probably due to the late elution of the latter.
Ferritin is similarly resolved on both phases. The basic decapeptide Bacitracin, appears to adsorb on the TSK (Kd 1.08) while eluting at a reasonable Kd of 0.91 on the Exsil.
Resolution of higher molecular weight BSA aggregates is slightly better on the TSK but the latter shows tailing and excessive retention of the basic Cytochrome C.
It appears that the TSK has slightly better resolution potential in the mid range while the Exsil has an extended high MW range potential and lower adsorptive effects with basic proteins.
Insulin and other proteins
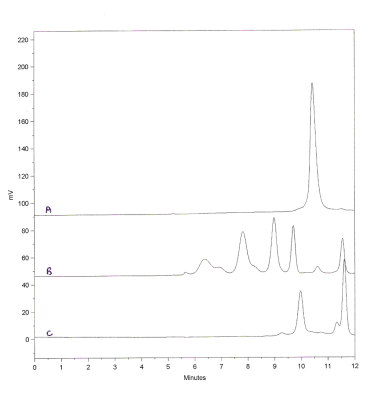
High Speed Separations
Although a number of phases are available for GFC of medium molecular weight proteins, they are either based on low pore volume 300A silicas which have too large a pore diameter to closely match TSK G3000SW, or like the latter, employ high pore volume silicas which have poor mechanical strength and typically have a maximum pressure limit of only 1000 psi. This is just adequate for the typical flowrates used (1 ml/min for 7.8mm id) as the pressure drop is around 700-800 psi. It does not however allow for any significant pressure buildup through use and precludes the use of higher flowrates for quick scan analysis.
Exsil GFC 3000 does not suffer these limitations and can be packed at 7000 psi. This allows the use of higher flowrates which provides faster analysis, reduces the time to equilibrate a new eluent and provides higher headroom for pressure increase during use. The latter can become an important issue for unattended operation.
Here Exsil completes the analysis in one third the time allowing rapid scanning of samples and selection of only those samples of interest which need to be run at a lower flowrate to enhance resolution.
In addition to increased speed, the high pressure stability allows multiple columns to be used while maintaining reasonable anlysis times.
Protein Recovery
Recovery was assessed using the acidic protein, Human Serum Albumin and the basic protein, Equine Cytochrome C using 50mM pH 7 potassium phosphate and 150 mM sodium chloride eluent. In neither case was there evidence of protein loss.
Phase Clean-up
Although the recommended working pH is 2 – 7.5, cleaning the column with 50 column volumes of 50% 10mM NaOH/50% MeOH had no effect on the column performance. If the clean-up step is restricted to 5 column volumes, then the column could be cleaned a minimum of 10 times. However, the low apparent protein adsorption would indicate that, provided samples and eluent are filtered, column clean-up should only be required infrequently.